Owren’s Disorder Market Size, Growth, and Forecast to 2035
Introduction
The Owren’s Disorder Market refers to the global industry focused on the diagnosis, treatment, and management of Owren’s disorder, also known as Factor V deficiency or parahemophilia. Owren’s disorder is a rare inherited bleeding disorder caused by a deficiency of coagulation Factor V, a protein essential for blood clot formation. Patients with this condition experience prolonged bleeding episodes, excessive bruising, and complications during surgery or trauma.
The market holds clinical importance due to the life-threatening nature of untreated bleeding disorders. Although Owren’s disorder is rare, early diagnosis and effective management are essential to prevent complications. The global healthcare system recognizes rare bleeding disorders as priority areas for research, orphan drug development, and specialized care.
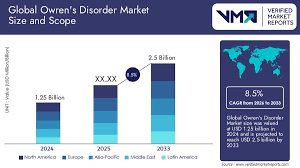
The Owren’s Disorder Market is valued at approximately USD 450 million in 2024. The market remains niche due to the low prevalence of the disorder, estimated at about 1 in 1 million individuals worldwide. Growth is supported by advances in diagnostic techniques, plasma-derived therapies, and rare disease funding programs.
Learn how the Owren’s Disorder Market is evolving—insights, trends, and opportunities await. Download report: https://www.databridgemarketresearch.com/reports/global-owrens-disorder-market
The Evolution
The understanding of Owren’s disorder dates back to 1947 when Norwegian physician Paul Owren first identified Factor V deficiency as a distinct bleeding disorder. Early treatment approaches relied primarily on fresh frozen plasma transfusions to replace deficient clotting factors.
In the following decades, improvements in coagulation testing allowed for accurate diagnosis of Factor V deficiency. Laboratory advancements in prothrombin time and coagulation factor assays enabled clinicians to differentiate Owren’s disorder from other bleeding disorders such as hemophilia.
The development of plasma-derived clotting factor concentrates represented a milestone in treatment evolution. These therapies improved safety and reduced reliance on whole blood transfusions. Enhanced screening protocols minimized risks of viral transmission.
Genetic testing technology further advanced the market. Identification of mutations in the F5 gene allowed for early diagnosis and family screening. The focus shifted from reactive treatment to preventive care and personalized management.
In recent years, interest in gene therapy and recombinant factor development has increased. Research initiatives targeting rare bleeding disorders aim to reduce treatment frequency and improve patient quality of life.
Market Trends
Several trends are shaping the Owren’s Disorder Market. Increased awareness of rare diseases is encouraging earlier diagnosis and referral to specialized hematology centers. Advocacy groups and rare disease networks are promoting patient education and genetic counseling.
Orphan drug development programs are influencing pharmaceutical research. Regulatory agencies provide incentives such as market exclusivity and expedited approvals for treatments targeting rare conditions. These incentives attract biotechnology companies to invest in innovative therapies.
There is a growing focus on personalized medicine. Genetic profiling and biomarker research support tailored treatment plans based on mutation type and severity of deficiency. Advances in molecular diagnostics enhance treatment precision.
Telemedicine and digital health platforms are improving patient access to specialized care. Remote monitoring and electronic health records support long-term management of rare bleeding disorders, especially in geographically isolated regions.
Regional adoption patterns show North America and Europe leading in advanced diagnostic capabilities and rare disease treatment programs. Asia-Pacific is expanding diagnostic awareness and healthcare infrastructure. Emerging markets are gradually improving access to coagulation testing and plasma therapies.
Challenges
The Owren’s Disorder Market faces several challenges. Low disease prevalence limits commercial incentives for large-scale drug development. Small patient populations result in limited clinical trial data and high per-patient treatment costs.
Access to specialized diagnostic facilities is restricted in low- and middle-income countries. Coagulation testing and genetic screening require advanced laboratory infrastructure, which may not be widely available.
High treatment costs present financial barriers. Plasma-derived therapies and hospital-based care can be expensive, placing pressure on healthcare systems and insurance providers.
Regulatory complexity for rare disease therapies increases development timelines. Clinical trials for rare conditions face recruitment challenges due to limited patient numbers.
Supply chain dependence on plasma collection creates vulnerability. Plasma-derived products rely on donor availability and strict safety protocols. Disruptions in plasma supply can affect treatment availability.
Market Scope
The Owren’s Disorder Market is segmented by treatment type, diagnosis method, end-user, and region.
By treatment type, the market includes fresh frozen plasma (FFP), platelet transfusions, antifibrinolytic agents, and investigational therapies such as recombinant factor products. Fresh frozen plasma remains the primary treatment option due to limited availability of specific Factor V concentrates.
By diagnosis method, the market includes coagulation testing, prothrombin time assays, activated partial thromboplastin time tests, and genetic testing. Genetic testing is gaining importance for early detection and family screening.
By end-user, the market includes hospitals, specialty hematology clinics, diagnostic laboratories, and research institutions. Hospitals account for the largest share due to emergency treatment requirements and transfusion services.
Regional analysis shows North America leading due to advanced rare disease programs, strong reimbursement policies, and active research initiatives. Europe follows with established healthcare infrastructure and orphan drug incentives. Asia-Pacific is experiencing growth due to expanding diagnostic services and healthcare modernization. Latin America and the Middle East & Africa are emerging regions with improving rare disease awareness.
Market Size and Factors Driving Growth
Several factors are driving growth. Increased awareness of rare bleeding disorders is leading to higher diagnosis rates. Improved laboratory testing capabilities enable accurate identification of Factor V deficiency.
Advancements in biotechnology and gene research are encouraging development of targeted therapies. Investment in recombinant clotting factors and gene-based treatments may improve long-term outcomes.
Government support for rare diseases is another key driver. Orphan drug incentives and funding for rare disease research stimulate innovation and market participation.
Rising healthcare expenditure in emerging markets supports improved access to specialized diagnostics and plasma products. Expansion of blood bank networks enhances treatment availability.
Population growth and improved life expectancy contribute to increased diagnosis of inherited conditions. Early screening programs and genetic counseling initiatives support preventive care.
Opportunities exist in gene therapy research and personalized medicine approaches. Collaborative research between biotechnology firms and academic institutions is expected to accelerate innovation.
Conclusion
The Owren’s Disorder Market represents a specialized segment within the global hematology and rare disease landscape. Despite its limited patient population, the market demonstrates steady growth driven by diagnostic advancements, rare disease policies, and therapeutic innovation.
Improvements in genetic testing, plasma safety, and investigational therapies are enhancing patient outcomes. Sustainable plasma supply chains and equitable access to treatment remain critical priorities.
Future opportunities lie in gene therapy, recombinant clotting factor development, and digital health integration. Stakeholders including pharmaceutical companies, research institutions, and healthcare providers are expected to benefit from ongoing innovation and increased awareness in the Owren’s Disorder Market.
Browse More Reports:
Europe Bakeware Market
Middle East and Africa Bakeware Market
North America Bakeware Market
Asia-Pacific Benign Prostatic Hyperplasia Devices Market
Europe Benign Prostatic Hyperplasia Devices Market
Middle East and Africa Benign Prostatic Hyperplasia Devices Market
Europe Bio-based Lubricants Market
North America Bio-based Lubricants Market
Asia Pacific Bioherbicides Market
Europe Bioherbicides Market
North America Bioherbicides Market
Asia-Pacific Bio-Implants Market
Europe Bio-Implants Market
North America Bio-Implants Market
Asia-Pacific (APAC) Biomarkers Market
About Data Bridge Market Research:
An absolute way to forecast what the future holds is to comprehend the trend today!
Data Bridge Market Research set forth itself as an unconventional and neoteric market research and consulting firm with an unparalleled level of resilience and integrated approaches. We are determined to unearth the best market opportunities and foster efficient information for your business to thrive in the market. Data Bridge endeavors to provide appropriate solutions to the complex business challenges and initiates an effortless decision-making process. Data Bridge is an aftermath of sheer wisdom and experience which was formulated and framed in the year 2015 in Pune.
Contact Us:
Data Bridge Market Research
US: +1 614 591 3140
UK: +44 845 154 9652
APAC : +653 1251 975
Email:- corporatesales@databridgemarketresearch.com
FAQ
1. What is Owren’s disorder?
Owren’s disorder, also known as Factor V deficiency, is a rare inherited bleeding disorder caused by insufficient levels of coagulation Factor V.
2. How large is the Owren’s Disorder Market?
The market is valued at approximately USD 450 million in 2024 and is projected to grow steadily through 2035.
3. What are the main treatment options?
Primary treatments include fresh frozen plasma transfusions, antifibrinolytic agents, and supportive care. Research into recombinant and gene therapies is ongoing.
4. Which region leads the market?
North America leads due to advanced healthcare systems, rare disease policies, and strong diagnostic infrastructure.
5. What challenges affect the market?
Key challenges include low disease prevalence, high treatment costs, limited clinical trial data, and dependence on plasma supply.
6. What is the future outlook for the market?
The market is expected to grow steadily with advancements in gene therapy, improved diagnostics, and increased global awareness of rare bleeding disorders.
- Art
- Causes
- Crafts
- Dance
- Drinks
- Film
- Fitness
- Food
- Games
- Gardening
- Health
- Home
- Literature
- Music
- Networking
- Other
- Party
- Religion
- Shopping
- Sports
- Theater
- Wellness


